Companies document development on blood checks to detect most cancers
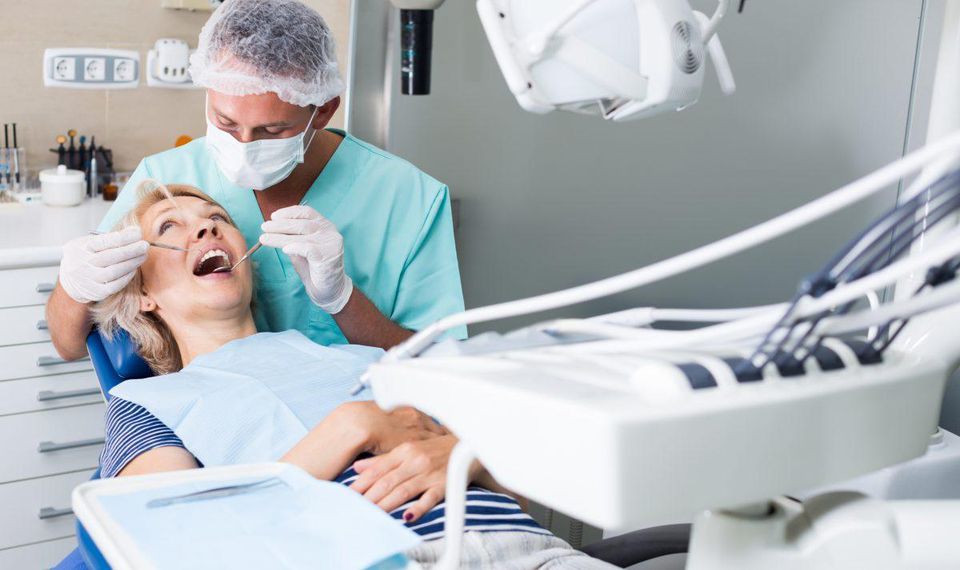
A California organization says its experimental blood took a look change capable of detecting cancer at an early degree and gave very few fake alarms to look at that included humans with and without the sickness. Grail Inc. Gave effects in an information launch on Friday and recorded them on Saturday at the American Society of Clinical Oncology meeting in Chicago. They have not been posted in a magazine or reviewed by different scientists. Many agencies seek to develop early detection “liquid biopsy” exams that seize bits of DNA that cancer cells shed into the blood.
On Thursday, Johns Hopkins University scientists released an organization known as Thrive Earlier Detection Corp. To develop its CancerSEEK, look at it, which yielded outcomes much like Grail’s greater than a year ago. Grail is intently watched because of the high-quality investment it’s attracted—more than $1 billion from Jeff Bezos, Bill Gates, and other celebrities.
The new effects blanketed 2,300 humans, 60% with cancer and 40% no longer acknowledged to have it. The check detected fifty-five % of regarded cancers and gave false alarms for 1%. The detection fee turned into higher—seventy-six%—for a dozen cancers that collectively account for almost two-thirds of cancer deaths in the U.S., together with lung, pancreatic, oesophageal, and ovarian.
The check found that about a third of cancers were at the earliest stage; however, as many as 84% had begun to spread, but now not extensively. It also cautioned that most cancers could occur in 94% of instances and become right approximately 90% of the time. That’s the most encouraging component because you do not need to tell humans they’ll have most cancers and then do many other checks to figure out which, said Dr. Richard Schilsky, leader clinical officer of the oncology society.
Dr. Len Lichtenfeld, interim leader clinical officer of the American Cancer Society, known as the low charge of fake alarms “terrific.” “They nevertheless have a long manner to head” to show the test’s worth as a screening tool; however, he said those effects are encouraging. “I do not doubt that in the next numerous years, we’re going to have a true early detection check.” However, the generation still needs to enhance and be tried in massive groups of humans without recognized cancers in which the detection rate won’t be as proper, he said.
The biggest query, he said, is “Will it make a difference in results?” Together with whether or not it facilitates human beings to live longer, the final degree of a screening check is well worth it. Grail’s review has not been compared to mammography, colonoscopy, or different screening equipment. It is not meant to update them, stated the organization’s chief scientific officer, Dr. Alex Aravanis. He noted many lethal cancers that the Grail takes a look at detected have no screening checks now.
It’s unclear what proof the U.S. Food and Drug Administration could require to not forget for approval. Sometimes, checks can be bought through looser lab accreditation pathways in place of via in search of FDA approval. Grail and Thrive have already got large studies underway. “We’re not going to diagnose every most cancer,” but won’t need to due to the fact so many aren’t determined now until it is too late for powerful remedy, said Dr. Minetta Liu, a Mayo Clinic cancer professional who’s supplying Grail’s outcomes at the most cancers conference.